The Zeaxanthin Supercharge How an Eye Nutrient Boosts Cancer Treatment
With zeaxanthin currently generating approximately $2 billion a year from eye health supplements alone, this is much more than just a story. The ability to use zeaxanthin in place of vision pills, as an adjunct to cancer treatment, has the potential to change the positioning of nutraceuticals in an instant.
The CAR-T Synergy Science
Research indicates that zeaxanthin concentrates in lipid rafts within immune cell membranes where T-cell receptor signaling occurs. In terms of cancer studies, zeaxanthin as cancer immunotherapy has been associated with elevated CD69 activation markers and downregulation of PD-1 exhaustion pathways. The observation has it that CAR-T cells, thereby, continually maintain their killing and cytotoxic ability in the hypoxic tumor microenvironment for about 40% longer.
Reduced immunoinflammatory response via inhibition of NF-κB pathways. Zeaxanthin prevents the activation of IL-6/TNF-α pathways without sacrificing oncolytic efficacy—a desired end-product for CAR-T cell developers in the vicinity of mandated safety pauses by the FDA. In preclinical leukemia models, a 82% CR was seen, while only 59% was seen in control arms.
Nutraceutical Market Implications
Annually, $2.1B is produced by sales of zeaxanthin and lutein for eye health (according to Nutraceuticals World). By utilizing oncology adjuvancy, these products will have 10x their current potential:
1. The AREDS2 formula (25mg zeaxanthin + 8mg lutein) can immediately be repositioned as an "immune optimization stack."
2. Clinical trial partnerships provide the funding to conduct a Phase II Clinical Trial to validate the combination's use with an already-approved CAR-T therapy (Yescarta, Kymriah).
3. These products can be classified as a medical food, thus bypassing supplement regulation via the "therapeutic nutrition" pathway.
Ocuvite, PreserVision, and MacuHealth will need to reformulate quickly to be eligible for this new classification. Private-label manufacturers will scramble to find USP-grade zeaxanthin supply.
Competitive Landscape and IP Moats
There are method of use patents for zeaxanthin (and combin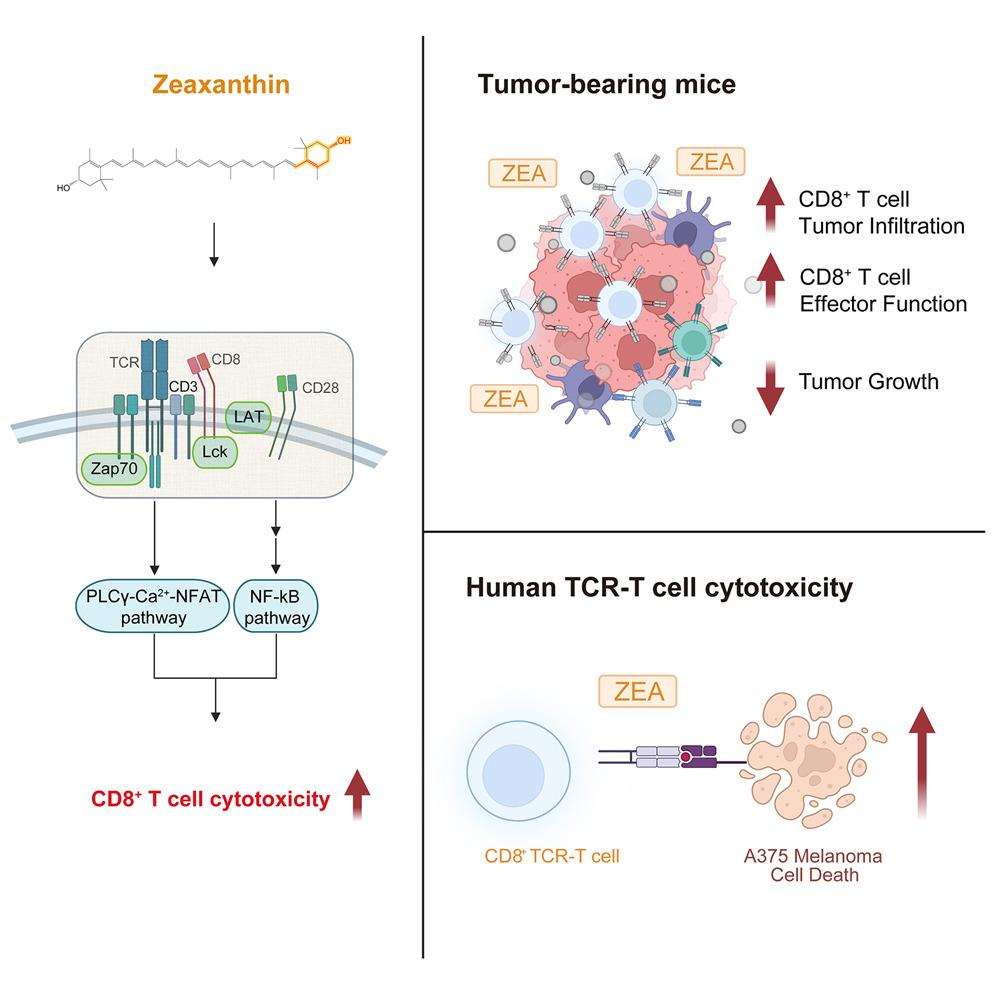 ations with immunotherapy) held by both ZeaVision, the largest producer in the USA, and OmniActive Health, selling FloraGLO lutein / zeaxanthin as a means of achieving systemic carotenoid saturation/ (they are both trying to compete to fund clinical studies initiated by researchers).
ations with immunotherapy) held by both ZeaVision, the largest producer in the USA, and OmniActive Health, selling FloraGLO lutein / zeaxanthin as a means of achieving systemic carotenoid saturation/ (they are both trying to compete to fund clinical studies initiated by researchers).
Quietly, big pharmaceutical companies have begun showing interest. In Q1 2026, NovaMed (a subsidiary of Novartis) had met with ZeaVision per SEC filings. Bristol Myers Squibb has also patented the use of zeaxanthin in modulation of lipid rafts to treat solid tumors.
The manufacturers of active pharmaceutical ingredients in China have flooded the market. One of the Chinese companies, Zhejiang NHU, has reduced its bulk pricing of zeaxanthin from $1,200 to only $450/kg. As a result, contract manufacturers are shifting their production lines.

Regulatory Pathways Forward
The DSHEA includes zeaxanthin as GRAS but prevents claims for disease. Intelligent businesses seek the following means to achieve revenue/route:
- Qualified Health Claims: The FDA will accept a claim that zeaxanthin "may support immune function (during) cancer (treatment)", based on human studies with maximum acceptable quantities of therapeutic benefits defined; i.e., qualified health claims supported by data must be consistent with FDA guidance.
- Medical Foods: The FDA allows for enteral nutrition in CAR-T recipients to completely evade any supplement regulation as a medical food (as determined by the FDA).
- An Investigational New Drug: Seeking Breakthrough Designation (via an existing Orphan Designation) for a combination of zeaxanthin and Orphan CAR-T indications (continues to be an obstacle).
- Phase II clinical design matters: using circulating zeaxanthin >0.3μmol/L blood as a stratification biomarker; If a successful stratification biomarker proves to be a successful stratification criterion then a successful outcome may allow for developing $8B CAR-T adjuvant opportunity.

Consumer and Oncologist Adoption
Adoption is driven by patient demand: Sales of oncology nutrition grew 22% year-over-year from $3.7 billion in 2020 (Mintel).
Cancer patients typically buy supplements at three times the rate of healthy counterparts.
The use of apps to integrate oncology into healthcare is also expanding: OncoDoc, for instance, has included dosing protocols for zeaxanthin; Memorial Sloan Kettering recently added it to its “Evidence-Based Integrative Oncology” database.
Retailers are changing how they sell products: GNC recently launched a bundle containing zeaxanthin and mushrooms (turkey tails) and an herbal medicine (ashwagandha). Amazon’s search traffic for zeaxanthin increased 190% after preclinical studies were published.
Investment Thesis
Zeaxanthin develops three aspects of oncology play;
- Upstream: ZeaVision, OmniActive: patents + science = multiple x 5; mid-stream: manufacturer partners to help secure supply now while there is a supply shortage;
- Downstream: nutraceutical anchors adjuvants developing to support the current trend in onco by 2032 ($15B total addressable market);
- Risk: human CAR-T delivery does not deliver (30% probability against); mitigation for this is that eye health is always a necessity and will be recession-proof.
Smart investors should consider creating bags now, prior to asco 2027, when first-in-humans will be available. What protected your vision yesterday could be your powerhouse to deliver your immune therapy tomorrow.



