The Protein of Aging Have Scientists Finally Found the Off Switch for Brain Decay
The researchers from UCSF have found that FTL1 protein is involved in brain aging; removing this protein from the brain of old mice will partially reverse their cognitive decline. This is the first and most compelling demonstration that modifying one molecular switch can regenerate lost neural connections and restore memory function. This presents an exceptional opportunity for healthtech investors, pharma executives and wellness brands to discover blockbuster solutions in fundamental science that are hiding in plain sight.
FTL1: The Brain's Silent Wrecking Ball
At UCSF, a group headed by Saul Villeda is looking at ferritin light chain 1 (FTL1), which is an iron storage protein, and how it works in the aging brain. Their studies have shown that FTL1 increases in the older mouse hippocampus compared to the younger mouse hippocampus and directly weakens synaptic connections between the two neurons. This results in poorer performance on memory tasks, more difficulty with spatial navigation, and slower learning.
The team was very surprised by the findings when they reduced levels of FTL1 in older animals. They found that synapses were able to regenerate after reduction of FTL1 levels, and that memory performance improved toward that of younger mice. Villeda said to Nature Aging that "this is more of a reversal than a delay" and that excess FTL1 in young genetically engineered animals can lead to early onset of menopause, which provides evidence that FTL1 not only correlates with but is causally related to aging.
Why This Breakthrough Actually Matters
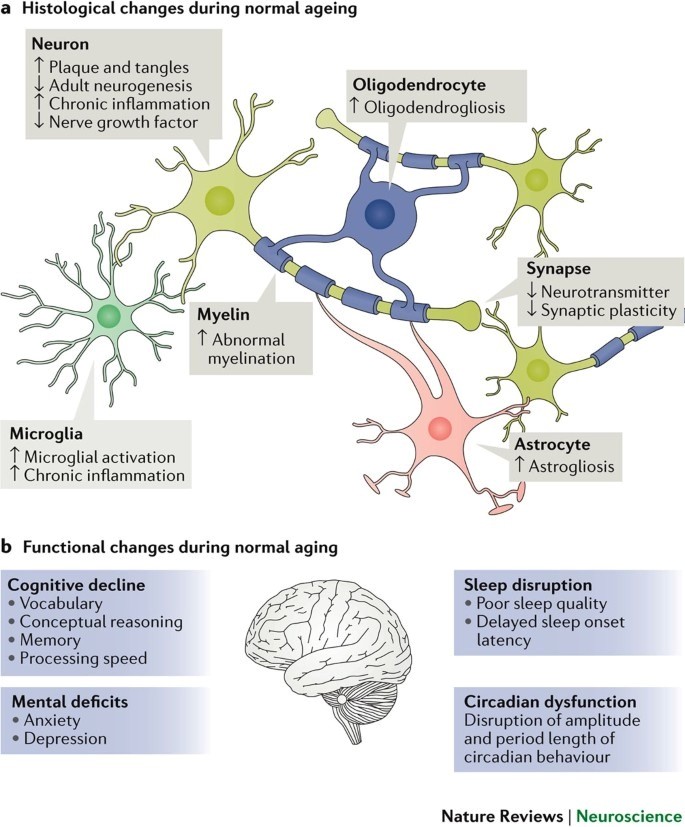
The majority of current aging research is symptom-related (to varying degrees) - e.g., studying plaque and tangles and whether or not they are present or caused by inflammation. FTL1 brain aging research is looking at the underlying cause of these issues: iron dysregulation in the brain that disrupts how our nerves communicate with one another (neural wiring). Ferritin normally stores free iron within the brain (to protect it from causing oxidative damage), but as our bodies age, ferritin has been shown to lose its ability to store free iron (in a healthy manner). Excess ferritin protein is thus preventing the proper transportation of free iron in the brain and overloading our synapses with free iron by preventing the normal "non-covalent" bonding between ferritin molecules, eventually resulting in lipid peroxidation and synapse loss.
This all applies as well from a business point of view. This body of research into iron metabolism can be useful as a base of evidence for researchers focus on iron as a way to possibly prevent or reduce the onset of neurodegenerative disease/disorders. Deferiprone, which is already FDA approved for patients with thalassemia, is being studied for its ability to chelate iron in the brain and for its potential to have a positive impact on patients with Parkinson's. In addition, FTL1-specific inhibitors or gene silencer could likely be leveraged for Alzheimer's disease, vascular dementia, and normal age-related cognitive decline or dementia.
The Market That's Already Moving
By 2030 neurodegenerative disease therapies will be a $50 billion+ market and there are many businesses that could take advantage of the FTL1 finding:
- Nutraceuticals/supplements - Heme iron blockers, lactoferrin, and IP6 (phytate) are already targeting brain iron and could be validated from clinical trials literally overnight.
- Precision diagnostics - FTL1 levels in blood or saliva will serve as biomarkers for cognitive aging and could be licensed from Quest/Labcorp now.
- Digital therapeutics - Apps with FTL1 genotyping as part of personalized dietary and iron protocols.
Faster than you can say "pharma," Biogen's iron homeostasis programs from 2022 are now suddenly looking very smart! Also, startups like Curasen (iron-Aβ clearance) will be pivoting hard.
Novartis' decision to invest further in their FTL1 iron dysregulation program two weeks after the UCSF study was published (by redistributing $120M from less exciting Aβ programs) coupled with Denali Therapeutics' news of their CRISPR-based FTL1 silencer entering preclinical studies (with human iPSC-derived neurons showing 28% recovery of synapses) and Quest Diagnostics filing for provisional patents for serum FTL1 assays to use as cognitive aging biomarkers means this isn't just interest in theory — this is a serious amount of money. When high-profile biotech companies are making major changes to their business strategy days after the release of a scientific paper, that constitutes a strong signal of interest. Iron homeostasis has now become the most active investment opportunity in the field of neurodegeneration (following alpha-synuclein).
Translation Hurdles (And How to Clear Them)
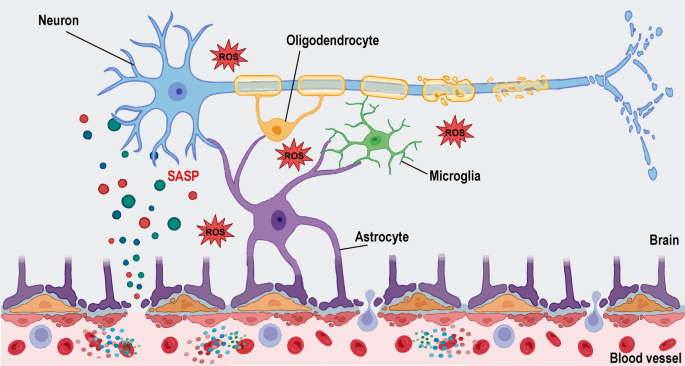
Tomorrow doesn’t mean human trials. The mouse hippocampus isn’t an exact substitute for either. We still need to confirm the contribution of FTL1 to pathology from Alzheimer’s (driving pathology with Aβ and/or tau). Inhibitors will have ongoing challenges with sufficient penetration for crossing the blood-brain barrier.
Smart money is hedged by first utilizing funding biomarker studies. If you find that serum FTL1 correlates with deterioration in cognitive function in individuals aged 65-80, you’ve successfully set FTL1 for the Phase 0 gold standard. To further enhance the Phase 0 biomarker studies as imaging endpoints, you would also include multimodal MRI using iron-sensitive sequences.
Because of a regulatory fast-track opportunity, there is potential for the FDA's 2025 Brain Health initiative, which prioritizes aging interventions based on mechanisms, to fast-track FTL1 modulators as the first approved class of agents for "Biology of Aging."
Investment Thesis: Three Plays
FTL1 has rewritten the guide for brain health:
- Biotech: Small Molecules/gene Therapy Targeting Ferritin Light Chain. Significant $200-$500 million Series A opportunity.
- There are testing kits using non-invasive FTL1 diagnostic tests and use to bill convenience CLIA laboratory facilities.
- Iron Optimized Brain Longevity Stacks; A Consumer Health Product.
The signal is loud and clear - Protein Dysregulation accounted for 70% of neurodegeneration. FTL1 is your lever to address this and those companies that define this biology will lead the way to the next decade of cognitive health.


